
- English
- Español
- Português
- русский
- Français
- 日本語
- Deutsch
- tiếng Việt
- Italiano
- Nederlands
- ภาษาไทย
- Polski
- 한국어
- Svenska
- magyar
- Malay
- বাংলা ভাষার
- Dansk
- Suomi
- हिन्दी
- Pilipino
- Türkçe
- Gaeilge
- العربية
- Indonesia
- Norsk
- تمل
- český
- ελληνικά
- український
- Javanese
- فارسی
- தமிழ்
- తెలుగు
- नेपाली
- Burmese
- български
- ລາວ
- Latine
- Қазақша
- Euskal
- Azərbaycan
- Slovenský jazyk
- Македонски
- Lietuvos
- Eesti Keel
- Română
- Slovenski
- मराठी
- Srpski језик
Differences between SiC crystals with different structures
2024-03-25
Silicon carbide (SiC) is a material that possesses exceptional thermal, physical and chemical stability, exhibiting properties that go beyond those of conventional materials. Its thermal conductivity is an astonishing 84W/(m·K), which is not only higher than copper but also three times that of silicon. This demonstrates its enormous potential for use in thermal management applications. SiC's bandgap is approximately three times that of silicon, and its breakdown electric field strength is an order of magnitude higher than silicon. This means that SiC can provide higher reliability and efficiency in high-voltage applications. Additionally, SiC can still maintain good electrical conductivity at high temperatures of 2000°C, which is comparable to graphite. This makes it an ideal semiconductor material in high-temperature environments. The corrosion resistance of SiC is also extremely outstanding. The thin layer of SiO2 formed on its surface effectively prevents further oxidation, making it resistant to almost all known corrosive agents at room temperature. This ensures its application in harsh environments.
In terms of crystal structure, SiC’s diversity is reflected in its more than 200 different crystal forms, a characteristic attributed to the diverse ways in which atoms are densely packed within its crystals. Although there are many crystal forms, these crystal forms can be roughly divided into two categories: β-SiC with cubic structure (zinc blende structure) and α-SiC with hexagonal structure (wurtzite structure). This structural diversity not only enriches the physical and chemical properties of SiC, but also provides researchers with more choices and flexibility when designing and optimizing SiC-based semiconductor materials.
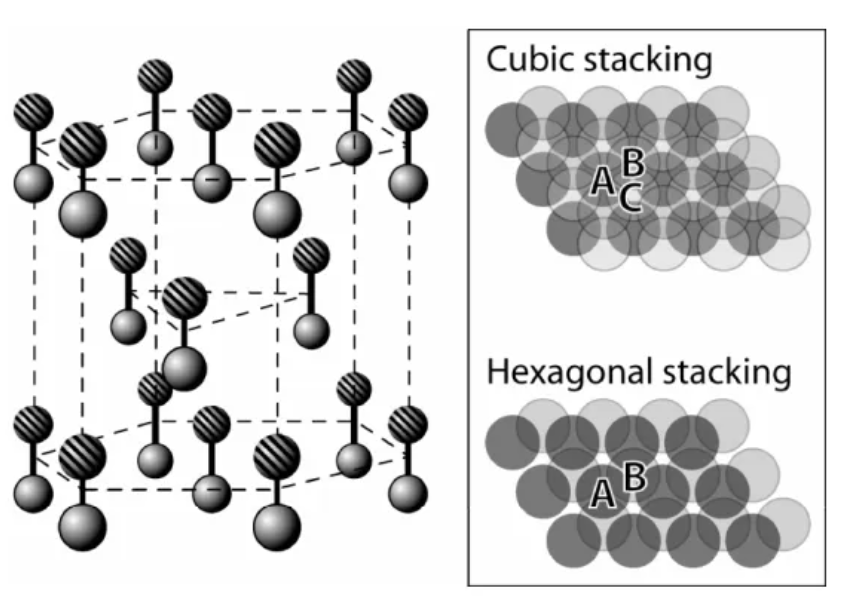
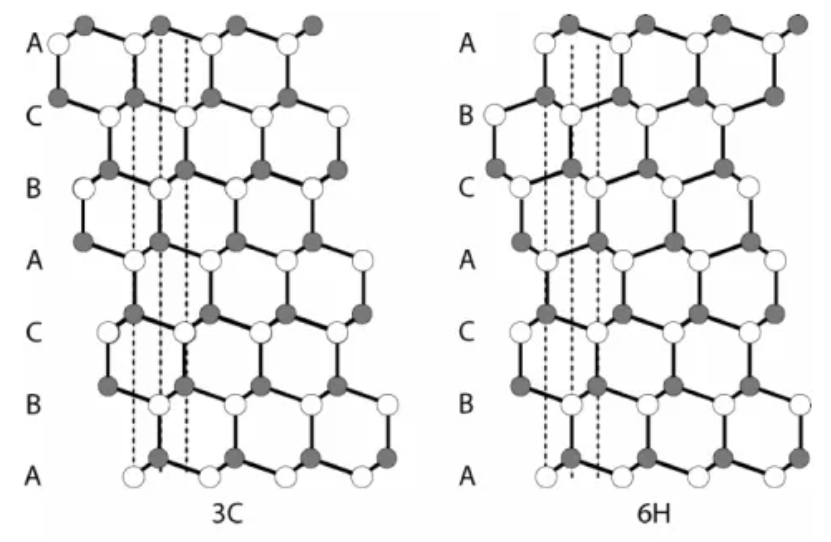
Among the many SiC crystal forms, the most common ones include 3C-SiC, 4H-SiC, 6H-SiC, and 15R-SiC. The difference between these crystal forms is mainly reflected in their crystal structure. 3C-SiC, also known as cubic silicon carbide, exhibits the characteristics of a cubic structure and is the simplest structure among SiC. SiC with hexagonal structure can be further subdivided into 2H-SiC, 4H-SiC, 6H-SiC and other types according to different atomic arrangements. These classifications reflect the way atoms are packed inside the crystal, as well as the symmetry and complexity of the lattice.
The band gap is a key parameter that determines the temperature range and voltage level in which semiconductor materials can operate. Among the several crystal forms of SiC, 2H-SiC has the highest bandgap width of 3.33 eV, indicating its excellent stability and performance under extreme conditions; 4H-SiC follows closely, with a bandgap width of 3.26 eV; 6H-SiC has a slightly lower bandgap of 3.02 eV, while 3C-SiC has the lowest bandgap of 2.39 eV, making it more widely used at lower temperatures and voltages.
The effective mass of holes is an important factor affecting the hole mobility of materials. The hole effective mass of 3C-SiC is 1.1m0, which is relatively low, indicating that its hole mobility is good. The hole effective mass of 4H-SiC is 1.75m0 on the base plane of the hexagonal structure and 0.65m0 when perpendicular to the base plane, showing the difference in its electrical properties in different directions. The hole effective mass of 6H-SiC is similar to that of 4H-SiC, but slightly lower overall, which has an impact on its carrier mobility. The effective mass of the electron varies in the range of 0.25-0.7m0, depending on the specific crystal structure.
Carrier mobility is a measure of how fast electrons and holes move within a material. 4H-SiC performs well in this regard. Its hole and electron mobility are significantly higher than 6H-SiC, which makes 4H-SiC Better performance in power electronic devices.
From the perspective of comprehensive performance, each crystal form of SiC has its unique advantages. 6H-SiC is suitable for the manufacture of optoelectronic devices due to its structural stability and good luminescence properties. 3C-SiC is suitable for high-frequency and high-power devices due to its high saturated electron drift speed. 4H-SiC has become an ideal choice for power electronic devices due to its high electron mobility, low on-resistance and high current density. In fact, 4H-SiC is not only the third-generation semiconductor material with the best performance, the highest degree of commercialization, and the most mature technology, it is also the preferred material for manufacturing power semiconductor devices in high-pressure, high-temperature, and radiation-resistant environments.




